Clinical trials involve extensive documentation that must be reviewed, approved, and archived with absolute precision. The Clinical Trial Document Approval Workflow, designed using Microsoft Power Automate and SharePoint, helps research teams manage protocols, consent forms, safety reports, and regulatory documents within a structured digital system. This workflow reduces manual coordination, strengthens audit compliance, and ensures research teams follow standardized approval steps required for trial accuracy and regulatory adherence.
Get a Free Demo of M365 based Approval Workflow
Approval Workflow Features
Customizable Approval
Automated Notifications
Audit Trail and Reporting
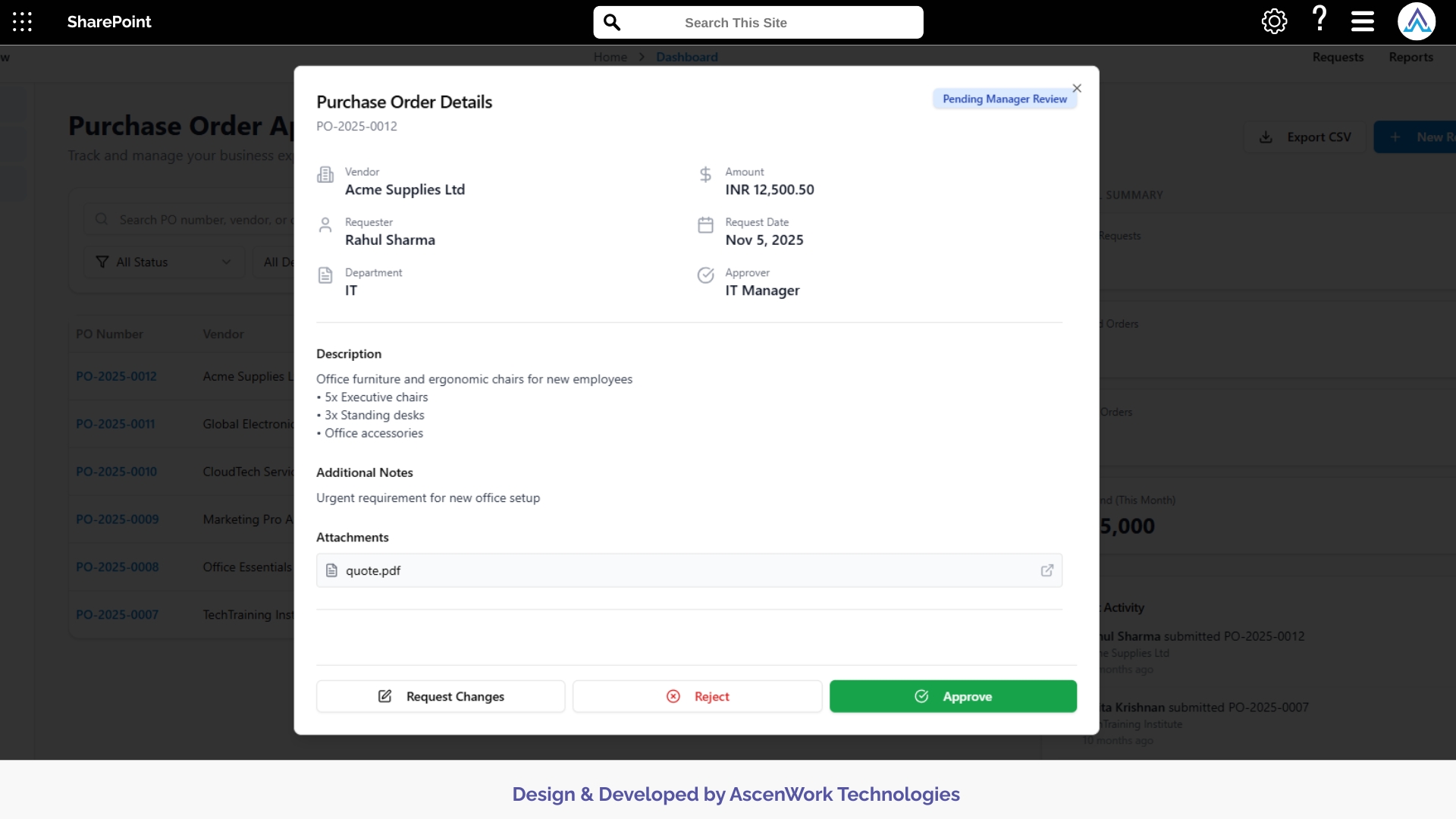
Approval Decision Options
Query Handling
Add Additional Approvers
Digitize and Streamline Your Clinical Trial Documentation Process
The Clinical Trial Document Approval Workflow enables research teams, investigators, ethics committees, and compliance departments to collaborate on trial documentation in an organized, trackable process. Built on SharePoint and Power Automate, the workflow ensures every document is reviewed in the correct order, meets regulatory requirements, and remains accessible for audits and monitoring.
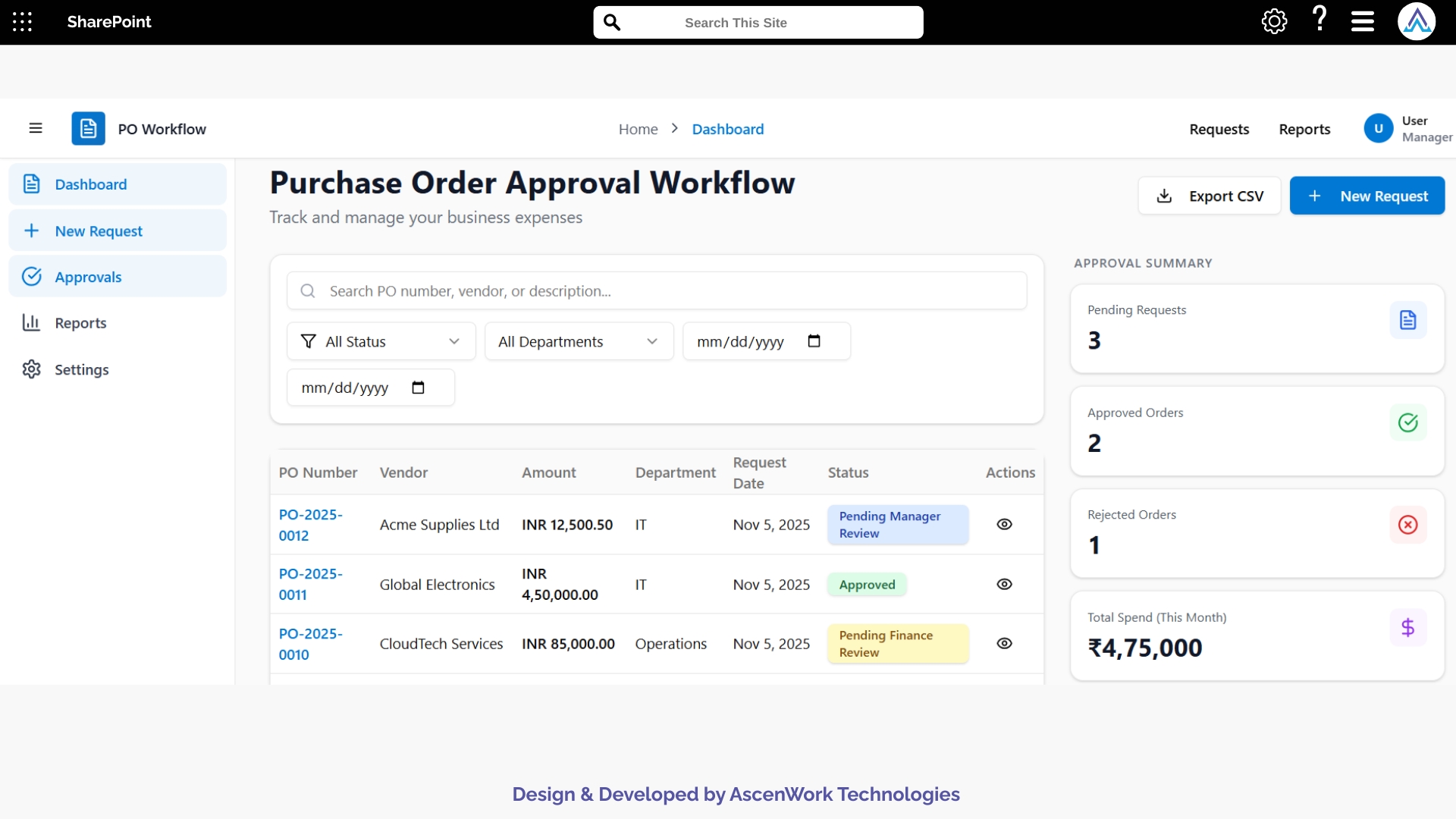
See how our approval workflow software automates your entire purchase order process in just a few clicks.
1. Centralized Document Submission Form
Researchers can upload trial-related documents using a dedicated SharePoint form that captures study details, document type, version number, submission date, and related protocol references. This ensures every document enters the workflow with complete context. Once submitted, automated routing begins, reducing manual follow-up and ensuring the correct stakeholders are notified for timely review.

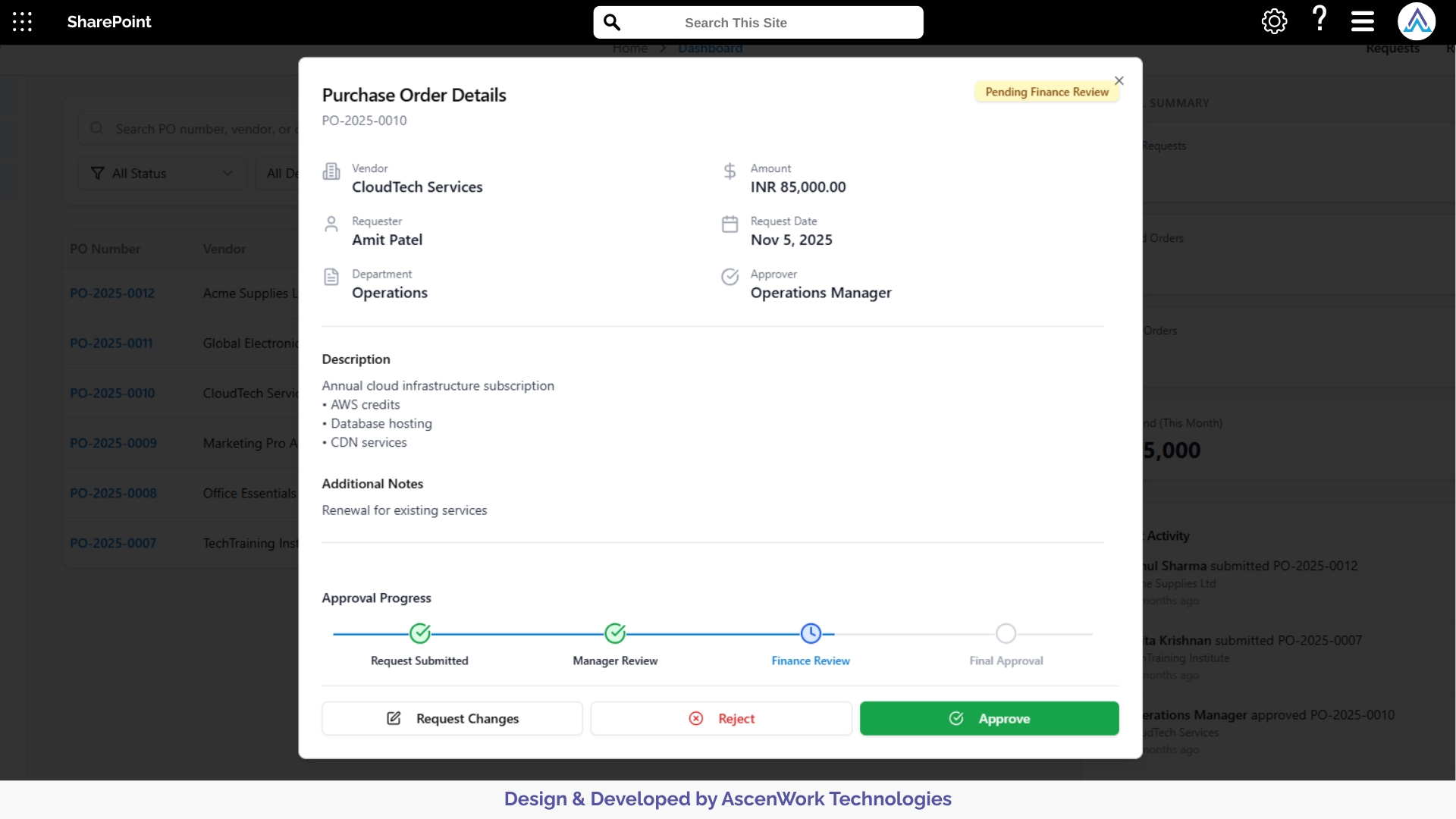
2. Multi-Stage Review and Approval Routing
Clinical trial documents often require sequential reviews from scientific investigators, quality teams, ethics committees, and regulatory leads. The workflow automatically directs documents to the appropriate reviewers based on the category and study phase. Each reviewer receives clear instructions, timelines, and document versions, ensuring smooth progression through each approval stage without miscommunication.
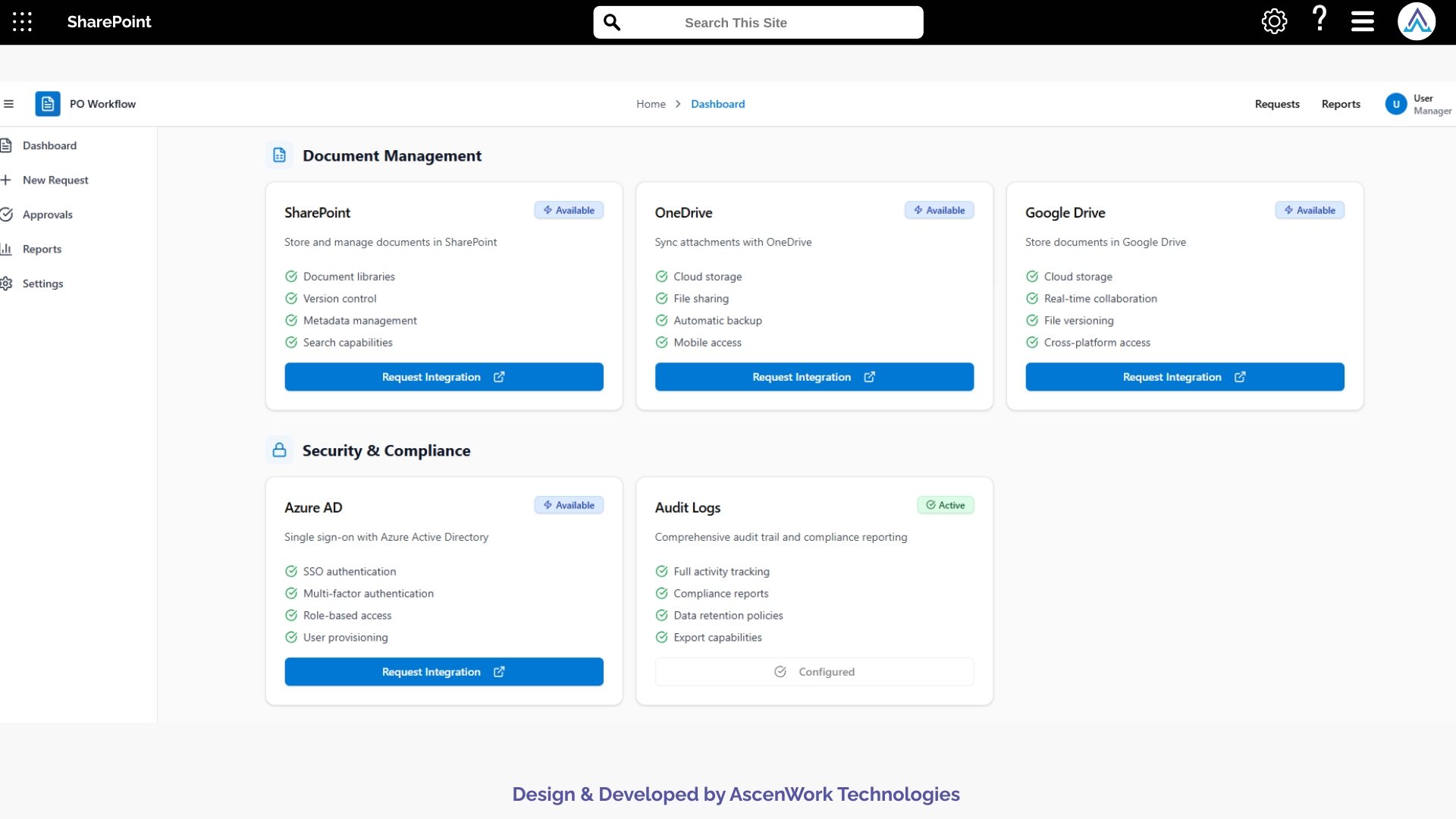
3. Regulatory and Compliance Verification
Before final approval, the workflow checks whether submissions meet required clinical research standards, including formatting rules, version control policies, and regulatory documentation criteria. Potential issues such as missing signatures, outdated versions, or incomplete forms are flagged for correction. This ensures documents remain audit-ready and comply with GCP, ICH, and institutional requirements.
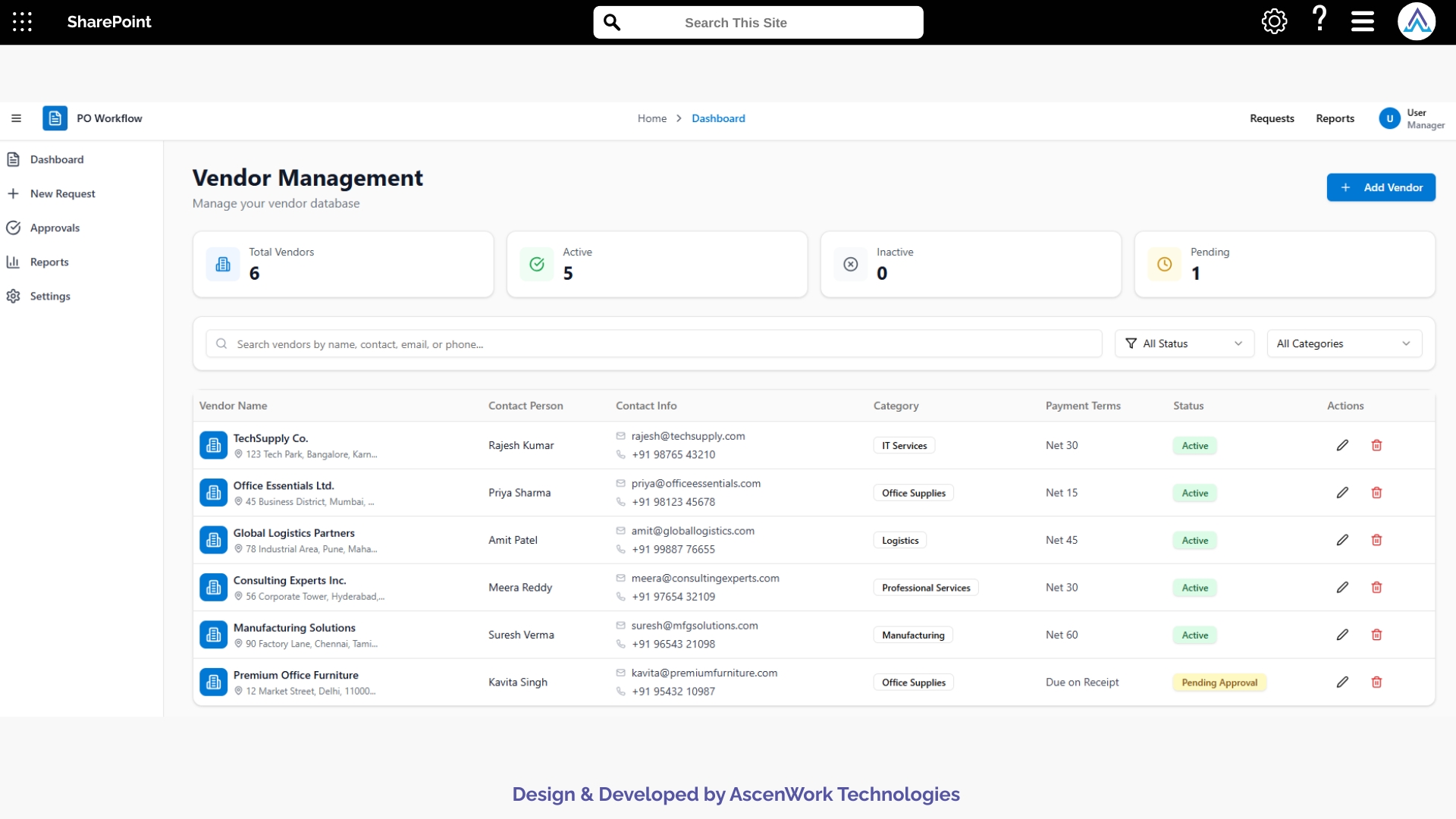
4. Continuous Alerts and Document Tracking
Stakeholders receive real-time notifications through Teams and Outlook whenever new documents arrive, require review, or need revisions. Automated reminders help prevent approval delays, which is critical for trial timelines. The tracking view provides teams with a complete overview of pending, approved, and revised documents throughout the trial lifecycle.
5. Secure Access Control and Document Permissions
Clinical research involves confidential data, requiring precise permission settings. The workflow assigns access based on study role—researchers, ethics committee members, regulatory reviewers, and documentation teams—each seeing only what they need. SharePoint’s granular security settings protect sensitive trial data and support compliance with institutional and regulatory guidelines.
Join leading finance and procurement teams using our workflow automation to eliminate bottlenecks and gain full control over spending.
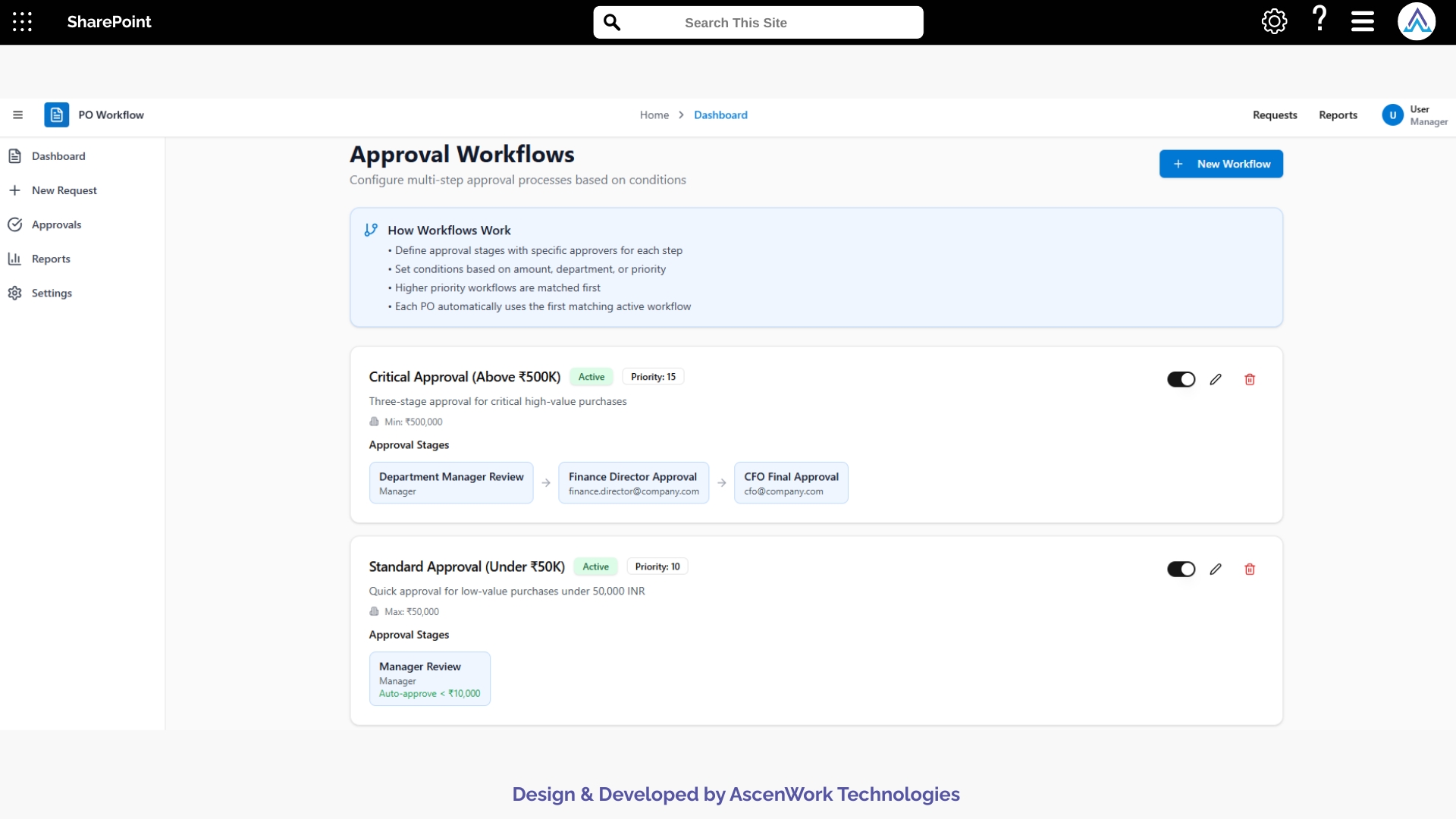
6. Analytical Insights and Document Metrics
Power BI integrations provide visibility into document turnaround times, reviewer workloads, compliance trends, bottlenecks, and study-specific approval durations. These insights help research coordinators predict delays, improve document quality, and ensure that trial milestones remain on schedule. Trend visualizations assist leadership in monitoring operational efficiency across multiple studies.
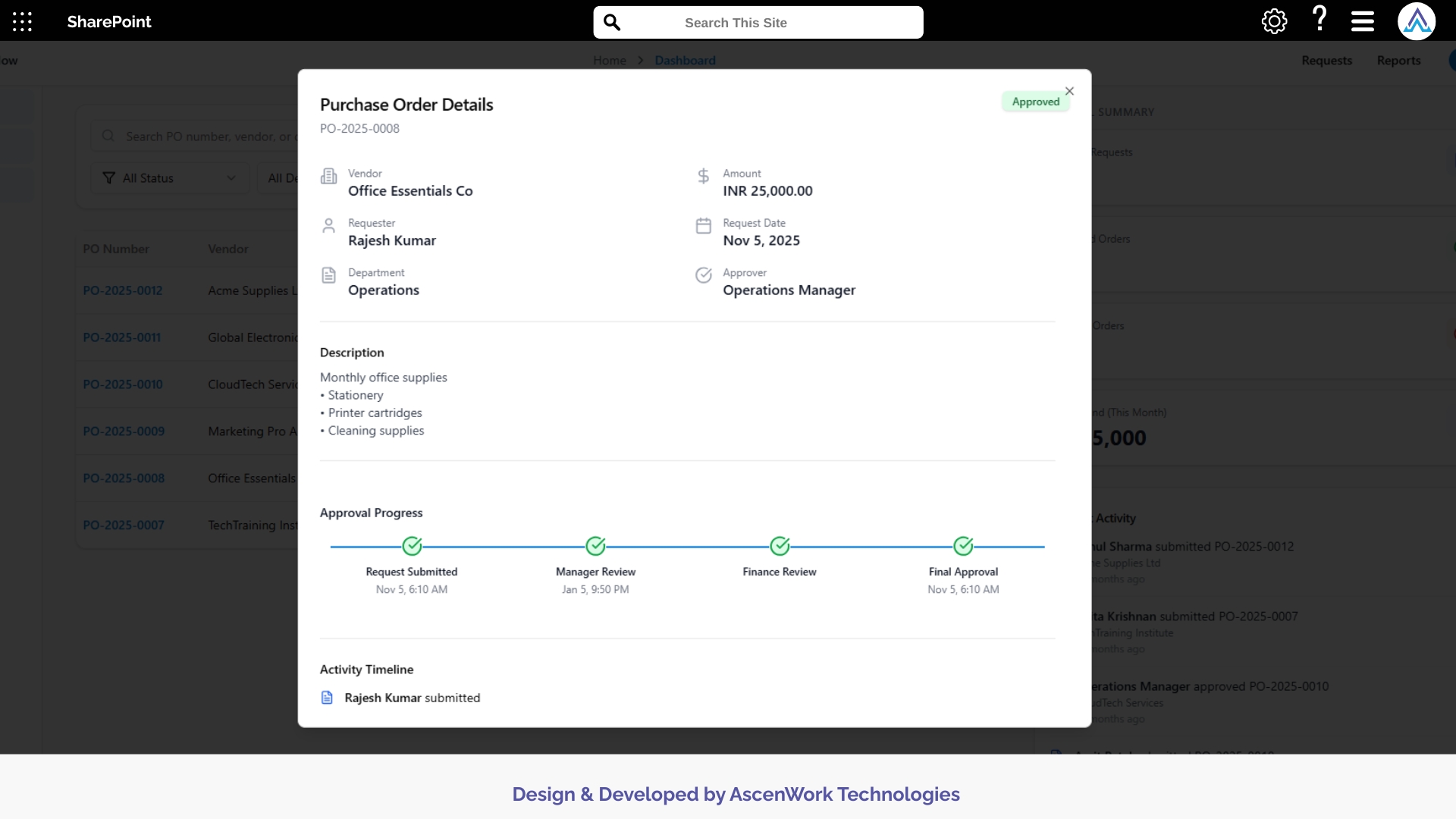
7. Complete Audit Log for Research Compliance
Every modification, comment, signature, and approval action is automatically logged. Audit records can be exported for ethics board reviews, regulatory inspections, and study sponsor audits. This detailed tracking supports transparency and ensures full traceability—essential for high-stakes clinical trials where documentation accuracy is mandatory.
8. Mobile-Enabled Review and Approval
Investigators and committee members can access documents, submit feedback, and approve revisions directly from mobile devices. Whether reviewing while traveling, on-site at trial locations, or working remotely, stakeholders can keep document progress moving without delays. Mobile access ensures flexibility and consistent momentum for time-sensitive studies.


Our consultants bring years of experience in automating purchase order and expense approval workflows across industries. We design solutions that fit your organization’s finance structure, ensuring faster approvals, accurate tracking, and full compliance with company policies.
Every business has a unique approval process. We build custom PO approval workflows that align with your departments, budgets, and hierarchy. From single-step to multi-level approval systems, our team ensures your workflow runs exactly the way you need.
Integrate your purchase order approval software directly with SharePoint, Power Automate, Teams, and Outlook. This lets finance teams manage everything from one place — automate repetitive steps, send reminders, and access audit data instantly within the Microsoft ecosystem.
Whether you manage a small finance team or a global enterprise, our solution scales effortlessly. We’ve implemented workflow automation software for organizations across multiple regions, ensuring consistent performance, data security, and accessibility from anywhere.
Our job doesn’t end at deployment. We provide continuous workflow monitoring, support, and optimization to ensure your PO approval process remains efficient. From new approval rules to user training, AscenWork is your long-term workflow automation partner.












AscenWork has built world class products on Microsoft 365
Have a question? check these answers.
Approval workflow software automates the process of reviewing and approving business requests — such as purchase orders, expense claims, or HR forms — ensuring transparency, accuracy, and faster decision-making.
Approval workflow routes requests automatically through predefined approval levels. Each approver gets instant notifications via email or Teams, and all actions are tracked within the system for full visibility.
Absolutely. Our approval software integrates natively with SharePoint, Power Automate, Outlook, and Teams, allowing your users to work within familiar Microsoft 365 apps.
Yes, administrators can modify the approver list anytime without interrupting existing workflows, making it flexible for organizational changes.
Your data is securely hosted on Microsoft’s trusted cloud infrastructure, with role-based permissions, encryption, and detailed audit trails ensuring full compliance and protection.
We offer prebuilt approval templates for common processes like purchase orders and HR requests, which can be customized further as per your business requirements.
Yes. The system sends automatic notifications to approvers and reminders for pending requests, ensuring no approval gets missed or delayed.
Implementation typically takes 2–4 weeks, depending on the complexity of your existing processes and integration requirements.
Finance, HR, Legal, Procurement, Manufacturing, and IT teams benefit the most — any process requiring sign-offs or compliance checks can be automated.
We offer both. AscenWork specializes in custom workflow development using SharePoint, Power Automate, and Power Apps to match your exact needs.
Yes. You can view detailed audit logs, approval histories, and performance analytics to ensure transparency and compliance during internal or external audits.
Yes, we provide video training, live workshops, and documentation so your team can confidently use the approval system from day one.
Yes. You can easily configure multi-level approvals, set conditions like amount limits, and define department-based approvers using our customizable workflow builder.
You can automate purchase orders, expense approvals, HR requests, vendor onboarding, document reviews, and more — all using the same platform.
Yes. Every request can be tracked in real time through your dashboard. Users can view who has approved, who’s pending, and the overall progress instantly.
Yes. Our workflows can be deployed both on Microsoft SharePoint Online or On-Premises, depending on your company’s IT policies and infrastructure.
Yes, we develop custom Power BI dashboards that visualize approval performance, pending requests, turnaround times, and departmental insights.
Because we combine Microsoft 365 expertise, custom development, and long-term support, ensuring you get a secure, scalable, and business-ready workflow solution tailored to your organization.
Yes. Our approval workflow software supports multi-department setups, allowing each department to have its own approval rules and processes.
Yes, we offer ongoing support, user training, and workflow optimization after deployment to ensure long-term success and user adoption.



